



Hani Alothaid, PhD, FIBMS
Al-Baha University
Reid Rubsamen, MD, MS, MHCM
CEO, Flow Pharma Inc.
Julie Lang, MD
Cleveland Clinic
M. Alshehri, PhD
Al-Baha University
Ahmed Alqahtani, PhD
KFSHRC
The long-term health, economic, and societal implications of long-COVID-19 are significant, and there is an obvious need to develop therapies to address and reduce the morbidity of this disease. FlowVax COVID-19 immunotherapy (FLOVID-20) works by teaching the patient's immune system to attack and kill virally-infected cells, potentially curing patients with Long Haul COVID.
The patented FlowVax platform uses tiny beads called microspheres—the same size as white blood cells—containing small protein fragments (peptides) of the COVID-19 virus. Once inside the body, these microspheres are collected by the cells of the immune system called macrophages. The FlowVax microsphere vaccine is 100% synthetic, does not contain animal products, and can be given as a single injection without requiring an intravenous infusion.
Download Full PDFSamples of FLOVID-20 for animal and human testing have already been manufactured. Flow Pharma has shown efficacy of FLOVID-20 in a Rhesus Macaque animal study conducted at the US National Laboratory at Galveston, Texas, and these results were published in a peer-reviewed journal. Flow Pharma has secured approval to test these samples in a human clinical trial which will be funded by this proposal.
The Centers for Disease Control and Prevention estimate that 1 in 13 adults in the United States have long-COVID-19 symptoms. As of May 2022, over 525 million confirmed cases of COVID-19 disease were reported worldwide, of which one in seven remained symptomatic at 12 weeks.
FLOVID-20 causes the immune system to attack the nucleocapsid protein at sites that have not mutated since the first appearance of the SARS-CoV-2 virus, suggesting that it will be effective against all future variants as well.
The retail price for PAXLOVID (without insurance) varies from $1,200 - $1,600 USD for a five-day course of therapy. If we price the required single injection of FLOVID-20 at $1000 USD/dose and assume 10% TAM penetration at launch (defining TAM as 10% of the estimated 1% of US adults with long haul COVID, or 300,000 people), we could achieve $300,000,000 gross revenue for the first year.
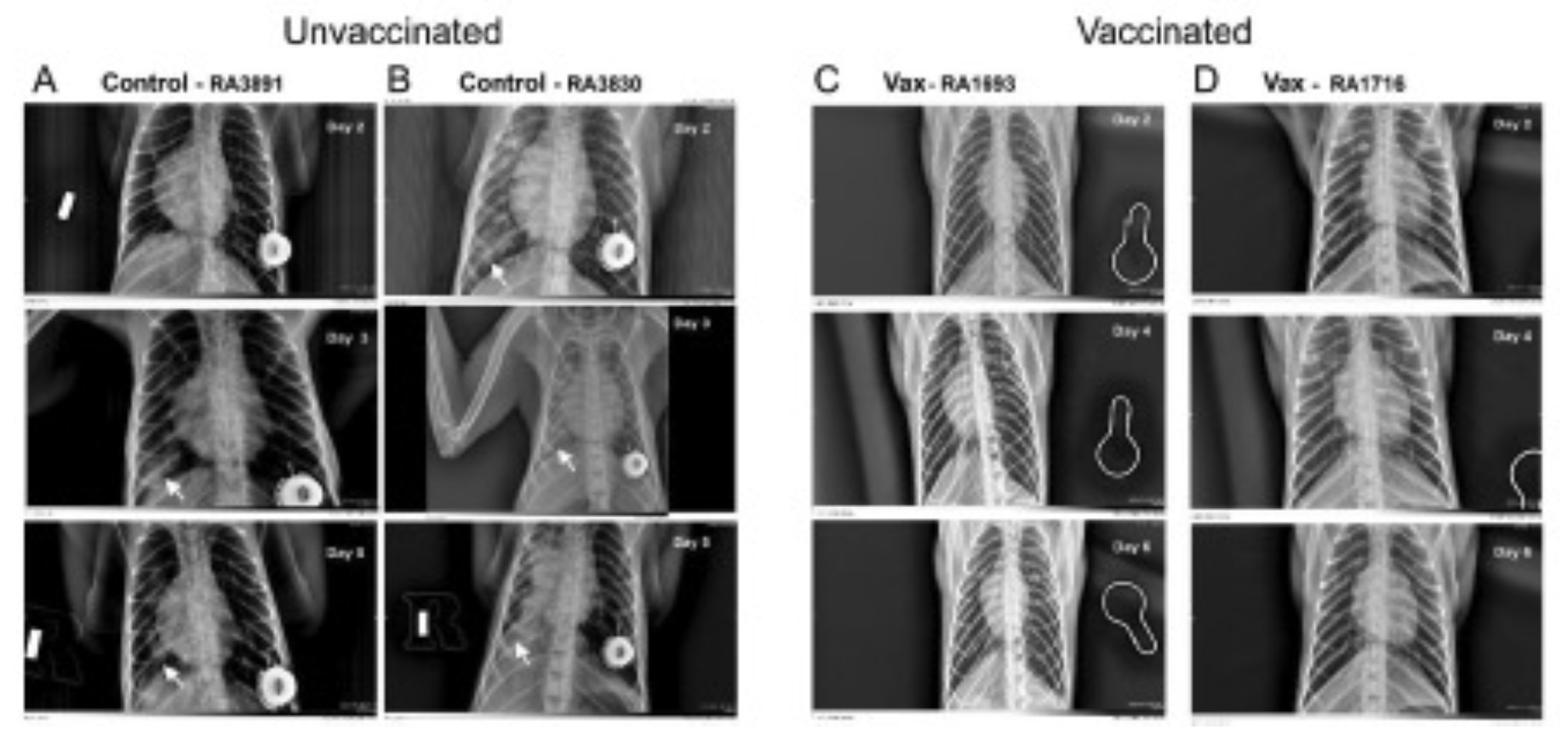
Figure 1: Representative chest radiographs of control and vaccinated macaques following SARS-CoV-2 challenge. Control macaques (left columns A and B) demonstrated a progression of pulmonary infiltrates during the acute period (Days 2-5) of disease post-challenge. In contrast, vaccinated macaques (right columns C and D) lacked similar abnormalities.
